Portola: Why I Think that AndexXa is Likely to be Approved (PTLA, Buy, $36.44)
Key Observations and Conclusions
- I believe there is a very strong possibility that AndexXa will be approved on its PDUFA date of May 4, 2018 even though the phase 3 trial that is the basis of the BLA leaves much to be desired. It is based on a single AndexXa arm which at this point has only enrolled 280 patients. This ordinarily would be what a phase 2 trial would look like. This modest data base has to concern the FDA and is the most significant issue that could lead the FDA not to approve the drug.
- I would expect a very sharp upward move in the stock if I am correct.
- However, AndexXa addresses the life threatening condition of excessive bleeding caused by anticoagulants that antagonize Factor Xa (notably Xarelto and Eliquis). There is no drug approved for this condition nor is there another drug in development on the near term horizon.
- Also on the encouraging side, the FDA has been moving aggressively to approve drugs addressing life threatening, unmet medical needs even without strong phase 3 data or even lacking phase 3 data. AndexXa fits perfectly into this paradigm.
- AndexXa appears to have a striking mortality benefit in treating excessive intracranial hemorrhages (ICH). Based on results in 139 patients presenting with ICH, the mortality rate for AndexXa is 12% as compared to a 43% mortality rate suggested by historical data. The benefit is less striking in non-ICH bleeds, perhaps 12% for AndexXa versus 15%.
- Although the FDA would clearly love to see results from a randomized controlled, clinical trial I see no chance of this. In the words of one opinion leader, no Internal Review Board at a major hospital would allow participation in a trial in which there is such a high mortality risk for ICH patients when Andexamet has shown (even though it is based on small numbers) a striking ability to sharply reduce mortality. It would be unethical to randomize some patients to a control arm.
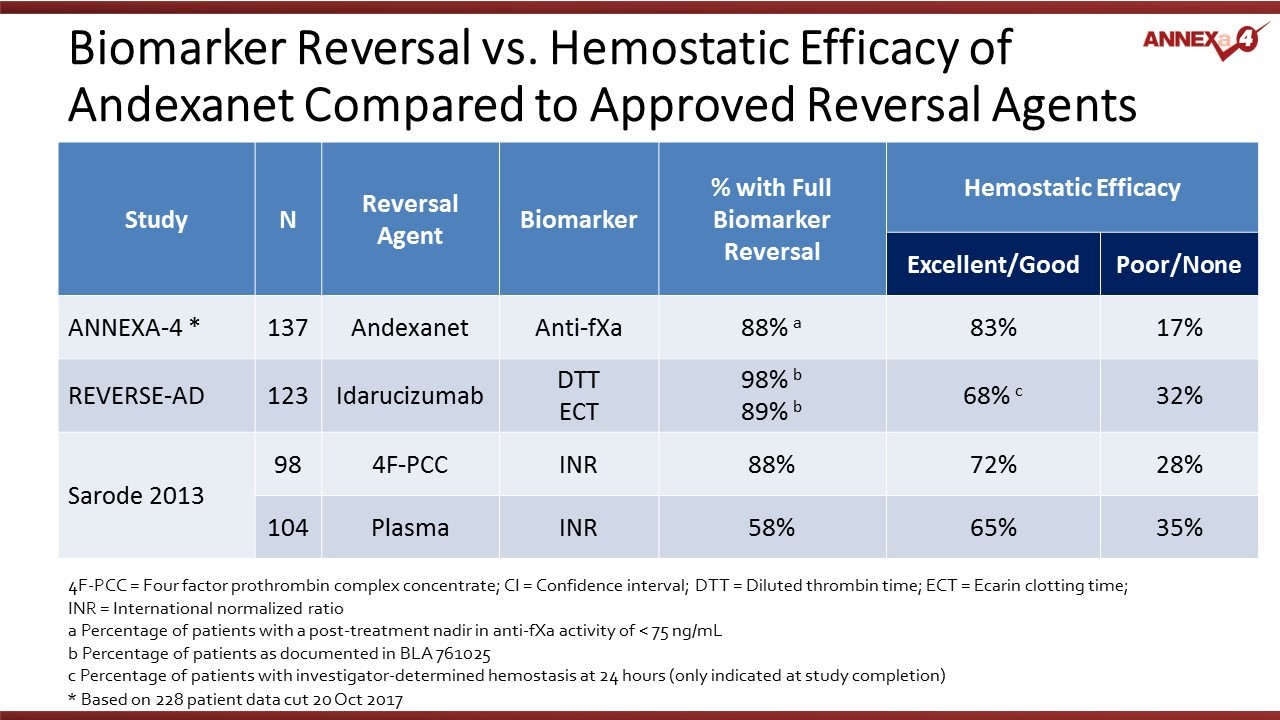
- Portola management indicated in its most recent investor call that the FDA seems to be focusing on a 5% disparity between the co-primary endpoints of full Factor Xa biomarker reversal (88% of patients) and good/ excellent hemostasis (83%) Portola points out that a similar (actually greater disparity of 20% to 30%) was seen in the clinical trial for Praxbind as a reversal agent for Pradaxa; note that the FDA approved Praxbind. Portola hypothesizes that this disparity is attributable to certain anatomy and physiology factors. I elaborate on this later in the report.
- If the FDA’s focus is primarily on this disparity, how could they apply a different standard to AndexXa approval than Praxbind? One potential reason is that the CBER division of FDA is reviewing the AndexXa BLA while CDER reviewed the Praxbind NDA. Hence different people are probably reviewing the AndexXa BLA.
- The FDA has informed Portola that the accelerated approval status for AndexXa remains in effect.
- I find that the medical condition in which the body’s clotting mechanism malfunctions and results in life threatening blood clots is hard to grasp. Likewise, a discussion of drugs used to treat this condition is esoteric and hard to understand. In the appendix of this report I have included a detailed discussion of these issues as seen by a layman (me). Other laymen may find this background analysis helpful.
Background for Report
This report addresses the potential for Portola’s AndexXa (andexamet) to be approved to reverse excessive bleeding that can result from the use of certain drugs used to prevent blood clots (popularly and wrongly referred to as blood thinners). AndexXa reverses the anti-coagulation effect of Factor Xa inhibitors, notably Xarelto (rivaroxaban) and Eliquis (apixaban) and also indirect Factor Xa inhibitors like Lovenox (enoxaparin). Xarelto and Eliquis were introduced in 2011 and 2012 respectively and have gained broad acceptance. In 2017, I estimate that worldwide sales of Xarelto were about $5.6 billion ($3.1 billion by Bayer and $2.5 billion by JNJ) and Eliquis sales were $3.5 billion). Lovenox and other low molecular weight heparins are also broadly used.
Xarelto and Eliquis have proven to be life saving drugs but their mode of action is to reduce the ability of blood to clot. In some cases this can lead to life threatening bleeding. Portola estimates that in 2017, there were approximately 100,000 hospital admissions in the US that were attributable to Factor Xa inhibitor induced excessive bleeding and the fatality rate was 15% to 20% (15,000 to 20,000 deaths). According to CDC estimates, this means that this is one of the top 15 causes of death in the United States. Portola’s AndexXa (andexamet) could be the first drug approved to address this important unmet medical need. The FDA has assigned breakthrough status to the drug, designated it for accelerated approval and set a PDUFA date of May 4, 2018. Until recently, the expectation of management and most analysts was that AndexXa would be approved in the US at the May 4, PDUFA date and introduced into the market soon thereafter.
Investors were stunned when management revealed during a recent conference call contents of a communication from the FDA received on February 23. This led to a 30% drop in the stock price. Let me elaborate. The regulatory filing for AndexXa is based on a non-randomized, single arm trial of AndexXa. Results will be compared to historical results achieved with standard of care. The FDA never officially agrees to any regulatory pathway, but the company and agency have discussed regulatory approval approach for several years and according to Portola, there was never any indication that the FDA would require a randomized trial for approval. Unexpectedly, the FDA indicated on February 23 that they “may” want data on some patients in a randomized study. If this is a condition for approval, the approval date for AndexXa in the US could be set back three years or so.
Analysis of More Recent Information on the FDA Situation
The majority of Wall Street analysts expected approval of AndexXa in the US on its PDUFA date of May 4, 2018. Portola management was as stunned by this new communication as investors and couldn’t offer much detail. However, they indicated that they might have more information that they could convey on a previously planned conference call to discuss updated results on AndexXa from its phase 3 trial at the American College of Cardiology Meeting on March 12, 2018. I spent several hours analyzing the slides presented on the call and listening to the discussion. I am not going to go into an in-depth analysis of these results. They were generally in line with results from earlier interim analyses. My intent was to see if management could provide additional information on what the FDA wanted; indeed this was the focus of most analyst’s questions. Here are the key takeaways from the call.
The ANNEXA-4 phase 3 trial has now enrolled over 280 patients and data was presented on 228 at the ACC meeting. These are patients adjudicated as having acute major bleeding. There are two co-primary endpoint:
- Change (hopefully reduction) in the anti-Factor Xa activity (a biomarker) when AndexXa is used to reverse excess bleeding caused by Xarelto and Eliquis.
- Clinical hemostasis efficacy through 12 hours (stopping bleeding by allowing clots to form). In the case of intracranial hemorrhage, this is determined with CT scans.
The slide presentation showed that AndexXa rapidly reversed anti-Factor Xa activity following a bolus of AndexXa. This was then followed two hours later by an intravenous infusion. The strong anti-Factor Xa activity was maintained over 12 hours. For major bleeds, an independent adjudication committer determined that 83% of patients achieved excellent or good hemostasis within a confidence interval of 76 to 89%. A 2014 survey of key opinion leaders indicated that 50% hemostasis would be clinically meaningful.
The effect of AndexXa is most striking in intracranial bleeds. Historical data from the 18,200 patient Aristotle trial indicated that 5% of patients treated with Xarelto had major bleeds. The mortality rate at 30 days for intracranial hemorrhage was 43% and for non- intracranial hemorrhage was 15%. In the ANNEXA-4 trial 227 patients were treated for major bleeds and because the trial was enriched to address intracranial bleeds, 139 of these patients were treated for ICH. The mortality rate in this 139 patient sub-group was a striking 12%. This appears to be the most clinically important attribute of AndexXa. Let me point out the obvious caveat that this is a small number of patients, but this is striking.
In the context of accelerated approval for AndexXa, the co-primary efficacy endpoints that the FDA is looking at are a biomarker that shows the degree of reversal of Factor Xa inhibition and hemostatic efficacy (forming clots to prevent bleeding). The following table shows results for AndexXa for both measures. It also shows similar measure for the REVERSE-AD trial that was the basis for approval of Praxbind (idarucizumab) for reversal of Pradaxa and the Sarode trial of 4F-PCC for reversal of warfarin.
Management was asked if they could provide additional information on what the FDA was looking for. They replied that the FDA seems most focused on a disparity between full biomarker reversal (in 88% of patients) and excellent/ good hemostatic efficacy (83% of patients). The question posed by the FDA is why some patients (around 5%) have full biomarker reversal, but poor or none hemostatic efficacy. Portola has responded in two ways.
Portola first pointed out that this disparity had also been seen in studies with two other reversal agents, Praxbind and 4F-PCC. The disparity for AndexXa was about 5% while for Praxbind it was 20-30% and for 4F-PCC it was 16%. There seems to be a consistent disconnect between the biomarker and the hemostatic efficacy. Portola also notes that Praxbind was approved with this data.
Management hypothesizes that this disparity may be due to anatomy and physiology. It is the case that patients not on anticoagulants will experience ICH and non-ICH bleeds. This may be because the lesions causing these bleeds are too large for the natural coagulation system to heal. Also, some of these patients may have high blood pressure to the extent that it complicates the ability of the hemostatic plug to hold.
Appendix
For those of you who would like additional information on the medial risk posed by abnormal blood clotting and drugs used to treat this condition, I have included the following discussion. It is written by a layman (me) in layman’s terms.
Background on Coagulation and Thrombosis (Blood Clots)
The blood clotting process (hemostasis) is essential to survival, but excessive clotting can lead to life threatening situations. When this occurs, it is necessary to give drugs which reduce the ability of blood to clot. These drugs are referred to as anticoagulants or anti-thrombotics. A blood clot is an aggregation of platelets (a type of red blood cell) within a blood vessel. Life threatening blood clots are usually caused by: (1) slowing of blood flow, (2) damage to a blood vessel wall that causes the generation of clotting factors, or (3) hypercoagulability arising from genetic disorders. Abnormal blood clot formation can block or partially block blood flow to tissues (ischemia) and this ischemia can cause tissue damage (infarction).
The most frequent medical problems caused by blood clots are:
- The formation of blood clots in the arteries of the heart can cause heart attacks.
- Blood clots that break off from vessel walls (usually veins in the legs), travel through the circulatory system and lodge in smaller vessels of the body; these are called thromboemboli. If they lodge in the brain they can cause strokes and if they lodge in the lungs they can cause pulmonary embolisms which impede the flow of oxygenated blood.
- Another major clot related problem stems from atrial fibrillation. This is an abnormal rhythm in the atrial chamber of the heart which causes a fluttering effect that results in a slowing of the blood flow. This can cause the formation of blood clots that then travel through the circulatory system into the brain and lungs.
The reasons for clots forming in the arterial system and the venous system are usually different. Arterial clots are often associated with plaque formation; the buildup of cholesterol and other lipids in the vessel wall that can rupture, cause clots to form and lead to ischemic disease such as acute coronary syndrome, stroke and peripheral artery disease. Cardiovascular procedures such as stenting can also damage vessel walls and lead to the formation of clots.
In this report, I will be discussing drugs used to treat clots arise in the veins due to slowing blood flow such as can occur in immobilized patients following surgery or hospitalization for other diseases and blood clots caused by atrial fibrillation which cause stasis or slowing of blood flow in the atrial chamber of the heart.
The Clotting Cascade Leads to Clot Formation
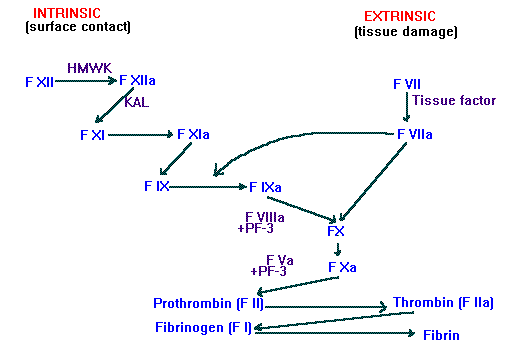
Anticoagulant drugs interact with proteins that interact with each other in a complicated clotting cascade that leads to formation of a clot. The essential cells in the clotting process are platelets which circulate in the blood. Platelets must be activated and aggregate at the site at which a clot is formed. Molecules on the surface of platelets then work in coordination with a second system that involves the actions of multiple proteins that are continually circulating in the blood called clotting factors which work together in a process that is known as the clotting cascade. As you will see shortly, this is an incredibly complex system. I will try to convey the basics of this system, but it is one in which I have only rudimentary understanding.
The essential function of the clotting cascade is to form fibrin, which wraps a mesh between the aggregated platelets to stabilize the clot. The clotting cascade involves numerous clotting factors, each of which has an active and inactive form. (The letter 'a' is used to denote an active form of a factor.) Following activation, one factor will then activate the next factor in a lengthy sequence until fibrin is formed. There are multiple interactions in the cascade that are required for coagulation to work properly.
The coagulation cascade takes place at the site where platelets have aggregated. The key steps are as follows:
- Tissue factor and factor VIIa activate factor X, forming factor Xa (remember the designation a means it is activated).
- Factor Xa is then able to activate factor II (also known as prothrombin) to form factor IIa (thrombin).
- Thrombin converts factor I (fibrinogen) to factor Ia (fibrin). Fibrin forms a mesh with the platelets that plugs the break in the vessel wall.
- The fibrin mesh is further stabilized by factor XIII, which sews up the clot (much like forming an intricate network of cross-stitched strands of fibrin).
- Factor V and factor VIII accelerate the conversion of factor X to factor Xa by factor IXa (this is done by factor VIII) and accelerate the conversion of prothrombin to thrombin as done by factor Xa.
This dizzying sequence is shown below:
Older Drugs Used for Treating Clots
Prior to 2010 when a number of important new drugs began to come to market, several older drugs were the backbone of therapy to treat or prevent clots. They were:
- heparin (unfractionated),
- the fractionated low molecular weight heparins (LMWH)
- warfarin which is generally referred to by the brand name Coumadin
- aspirin and Plavix (clopidrogel)
Heparin is a naturally-occurring anticoagulant produced by mammalian cells that is harvested from the intestines of pigs. It acts by preventing the formation of clots. It does not work directly to break down clots that have already formed; instead, it allows the body's natural clot dissolving mechanisms to work normally to break down clots that have formed. Heparin needs to be given by continual infusion and this means that it is generally used for short term anticoagulation in the hospital setting for treating acute coronary syndrome, atrial fibrillation, deep vein thrombosis and pulmonary embolism, cardiopulmonary bypass for heart surgery, and extracorporeal life support and hemofiltration. It requires careful monitoring to make sure that correct amounts are given.
The low molecular weight heparins (LMWH) are comprised of smaller (fractionated) sub-segments of the heparin molecule. Some of the better known LWMH products are Lovenox (enoxaparin) and Fragmin (dalteparin). They can be given subcutaneously and unlike heparin do not require continual monitoring. Because LMWH drugs have more predictable pharmacokinetic and anticoagulant effects, LMWH is recommended over unfractionated heparin for patients with massive pulmonary embolism and for initial treatment of deep vein thrombosis.
Prophylactic treatment of hospitalized medical patients with LMWH results in a significant reduction of risk for symptomatic deep vein thrombosis. Because it can be given subcutaneously and does not require monitoring, LMWH can be used as outpatient treatment in conditions such as deep vein thrombosis or pulmonary embolism unlike heparin which can only be given in a hospital. However, the need to give one or two subcutaneous injections per day limits effective utilization in the outpatient setting.
Warfarin or Coumadin (the brand name of the most widely used warfarin product) was long the standard of care in treating clots that form from atrial fibrillation. Frequently, patients requiring long term anticoagulation are started on LMWH and then switched to oral warfarin. Warfarin inhibits the synthesis of clotting factors in the clotting cascade that are biologically dependent on vitamin K. It is a difficult drug to use. It requires tight monitoring to titrate to the correct dosage and even after the right dosage is determined, the patient must have blood drawn every two weeks to make sure that the dosage remains correct. It has frequent and troublesome interactions with food and other drugs. Like LMWH it has a slow onset of action of a day or so.
Aspirin and Plavix (clopidrogel) are oral agents that are the mainstays of managing arterial clots. They are used prophylactically to reduce the risk of heart attacks. These drugs works on different biological targets: aspirin on the COX-1 enzyme and Plavix on the P2Y12 receptor on platelets. Both mechanisms prevent the aggregation of platelets that is necessary to form blood clots.
Newer Anticoagulants
The older anticoagulants played a critical role in cardiovascular medicine saving countless lives each year and were reasonably safe. However, there remained an unmet need for new products that could be given orally to extend treatment for which the injectable formulations of heparin and LMWH are not practical. There was also a need for new products as effective as warfarin, that didn’t have all of the dosage and monitoring issues of this drug. This opened the door to newer drugs like Pradaxa (dabigatran), Xarelto (rivaroxaban), and Eliquis (apixaban) and Savaysa (edoxaban). Pradaxa is a direct thrombin inhibitor. Xarelto, Eliquis and Savaysa are factor Xa inhibitors that work in the clotting cascade upstream of thrombin, which needs factor Xa for its formation. Both approaches reduce the amount of thrombin formed and consequently the production of fibrin.
The time of US approvals for preventing strokes due to atrial fibrillation for these three drugs were as follows:
- Pradaxa-October 19. 2010
- Xarelto-November 4, 2011
- Eliquis-December 28, 2012
- Savaysa-January 8, 2015
The time of US approvals for treating deep vein thrombosis and pulmonary embolism were as follows:
- Xarelto-November 2, 2012
- Pradaxa-April 7, 2014
- Eliquis-August 21, 2014
Other approved indications were:
- Eliquis- for reducing the risk of clots following hip or knee surgery March 18, 2014
- Pradaxa-Prophylaxis of deep vein thrombosis and pulmonary embolism after hip replacement surgery November 23, 2015
One of the major advantages offered by these new drugs was that they were just more convenient to use. Drugs are only effective if patients use them correctly and this was the problem with older drugs. The need to infuse heparin limits its use to the acute hospital setting. While low molecular weight heparins can be used outside the hospital, one or two injections per day is a difficult regimen for patients to adhere to. Coumadin (warfarin) is just a royal pain in the neck to initially titrate and then maintain at the right dosage. Pradaxa, Xarelto, Eliquis and Savaysa are once or twice a day oral drugs that are as effective as low molecular weight heparin or warfarin but require no significant titration, no monitoring, have few food and drug interactions and have a fast onset of action.
Antidote for Factor Xa Drugs
The entry of factor Xa inhibitor drugs has led to broad usage as they reduced the monitoring and dosing issues associate with warfarin and the dosing issues with LMWH. While they were an important advance in anticoagulation therapy, they presented a major drawback in that there was no antidote to reduce major bleeding episodes that occur when patients are on the drugs. Hence, many physicians and patients still perceive warfarin as safer because there are reversal strategies for when major bleeding occurs. The need for an antidote for the factor Xa inhibitor drugs is critical. Portola has developed AndexXa (andexamet) as a way to reverse the effects of the factor Xa drugs. AndexXa has a pronounced first mover advantage.
In October 2015, Praxbind (idarucizumab), a humanized monoclonal antibody antigen-binding fragment that binds to Pradaxa (dabigatran), received expedited approval from the Food and Drug Administration for use in the United States. This drug is an effective antidote for Pradaxa, but not for the factor Xa inhibitors.
Tagged as AndexXa approval on May 4, Portola + Categorized as Company Reports, LinkedIn