Neuralstem: Dr. Eva Feldman’s Latest Presentation on The Use of CUR’s Stem Cells in ALS (CUR, Buy, $4.04)
Overview
Dr. Eva Feldman is the lead investigator for the clinical trials of Neuralstem’s (CUR) NSI-566 spinal cord stem cells in the treatment of amyotrophic lateral sclerosis (ALS or Lou Gehrig’s disease). She spoke at the Keystone Symposia on April 9, 2014. Dr. Feldman is the recent President of the American Neurological Association. She is a Director of the A. Alfred Taubman Medical Research Institute and Director of Research of the ALS Clinic at the University of Michigan Health System. Dr. Feldman is an unpaid consultant to Neuralstem.
The Keystone Symposia on Molecular and Cellular Biology is a 501(c) (3) nonprofit organization with a 42-year history of convening open, peer-reviewed conferences that connect the scientific community and accelerate life science discovery. Dr. Feldman updated results for the phase 1 trial in ALS and had some preliminary comments about the phase 2 trial now underway. Here is the link to her slide presentation:
http://investor.neuralstem.com/index.php?s=126&item=149
I know that I am supposed to put my conclusions up front so that readers can quickly get an idea on my conclusions. However, I think that this might be confusing if you don’t understand the design and results of the trial in some detail. Hence, my conclusions are at the back of this report.
The Design of the Phase 1 Trial
The phase 1 trial can be a bit complicated so I will try to summarize its design. The trial enrolled 15 patients in total.
- Cohort A: The first six patients enrolled were given five unilateral (on one side of the spine) injections in the lumbar (lower) region of the spine. The injections contained 100,000 cells; all injections in the trial had the same number of cells. These were patients 1, 2, 3, 4, 5, and 6 and all were advanced and very sick non-ambulatory patients.
- Cohort B: The next 3 patients treated also received five unilateral injections in the lumbar region of the spine. These were patients 7, 8 and 9 and they were healthier than Cohort A patients and ambulatory at the time of transplantation.
- Cohort C/E: The next three patients were also healthier ambulatory patients who were given ten bilateral (on both sides of the spine) injections in the lumbar region. They were then brought back 1 ½ years later for a second surgery in which they received five unilateral injections in the cervical (upper) region of the spine. They are referred to in this presentation as patients 10, 11 and 12. Ted Harrada was patient 11.
- Cohort D: These patients were also ambulatory patients. They were suffering from bulbar symptoms that are related to deterioration in nerves in the cervical regions of the spine and control functions such as breathing, eating and speech. ALS symptoms usually start with deterioration of nerves in the lumbar region and then then progress over time to the cervical region. These three patients were the only patients in the trial with bulbar symptoms (I will explain the importance of this later.) They received five unilateral injections in the cervical region. These were patients 13, 14 and 15.
Updated Results from Phase 1 According to Cohorts
The primary endpoint of this study was the ALSFRS-r scale. The FDA has accepted this measure as a validated endpoint for ALS trials. If you would like a refresher on ALSFRS-r please refer to the appendix of this report. I will use ALSFRS-r scores extensively to interpret slides presented by Dr. Feldman in her studies.
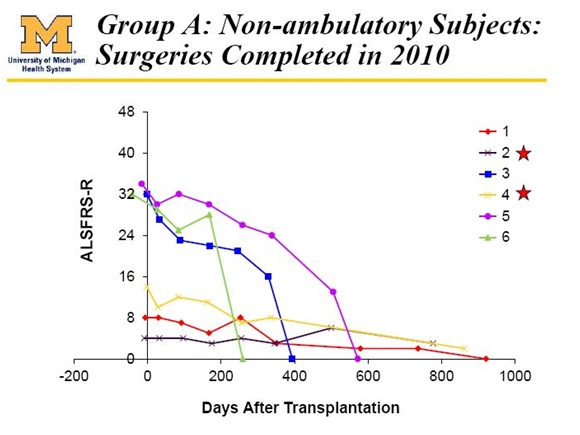
The First Cohort; Group A
The study started with six non-ambulatory patients with advanced disease. The FDA was concerned in phase 1 about safety for both the neural stem cells and the surgery used to implant the cells and required the trial to progress very cautiously and in patients with short life expectancies. There was little hope that these advanced patients could benefit; the intent was to demonstrate safety of the cells and surgical procedure. The ALSFRS-r results in these first six patients are shown below.
Let me try to put this in some perspective. For a normal person, the ALSFRS-r score is 48. Most ALS patients are diagnosed at an ALSFRS-r score of 32 to 36. Patients 3, 5 and 6 were non-ambulatory with an ALSFRS-r score of 32 to 34 at transplantation. They declined fairly quickly and died in 1.6, 1.1 and 0.7 years after transplantation. Patient 5 did the best and their ALSFRS-r score declined 32 points over about 20 months or a rate of 1.6 points per month. Historical experience suggests that ALSFRS-r should decline at about 0.9 points per month so it appears that patients 3, 5 and 6 had a severe disease course.
Patients 1, 2 and 4 had very low ALSFRS-r scores of about 5, 8 and 14 when transplanted. These very low scores usually occur at the time of death. The reason for these low and sustained scores is that the patients’ breathing was being maintained by mechanical ventilators. The three ALSFRS-r ratings that pertain to breathing were not applicable; taken off ventilators these patients would immediately die. They were also partially paralyzed and hence ALFFRS-r scores related to some motor functions also couldn’t change.
In any event, none of these six patients appeared to receive any benefit from the transplantation. This was as expected and is not surprising. The purpose was to demonstrate safety of the cells and the surgery in a group of very sick, advanced patients.
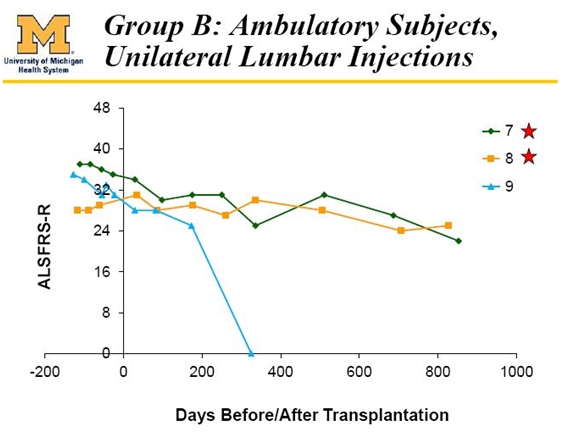
Cohort B
The next three patients were given five unilateral injections in the lumbar region of the spine. These were ambulatory patients who were healthier and less advanced than those from Cohort A. One of the first three patients, patient 9, died from a heart attack that the investigator concluded was unrelated to the disease or the treatment.
Patient 7 was transplanted with an ALSFRS-r score of 35 and twenty seven months later had an ALSFRS-r score of 23. Historical studies suggest that ALSFRS-r declines at about 0.9 points per month so that based on this I would have expected an ALSFRS-r score of 11 after twenty seven months and for the patient to be near death.
Patient number 8 had an ALSFRS-r score of 31 at transplantation and 25 nearly twenty six months later. Based on historical experience, I might have expected an ALSFRS-r score of 8 and for that patient also to be near death.
It appears that patients 7 and 8 benefitted to some degree from the transplantation. I asked Richard Garr, CEO of Neuralstem about these patients. He said that patients 7 and 8 are doing well and patient 7 almost two years post-surgery began to get better. When Mr. Garr saw him last October 17 in Atlanta he actually could get out of his wheel chair and stand and walk a bit for the first time in a year or more. The occasion was a fund raiser for ALS. Patient 7 asked members of the audience to raise their hands to pledge $1000 if he could get out of the wheelchair which he then proceeded to do.
Mr. Garr said that this is very encouraging data. All the focus has been on Ted Harrada’s cohort, but it’s clear that the cells have had an impact in this cohort as well.
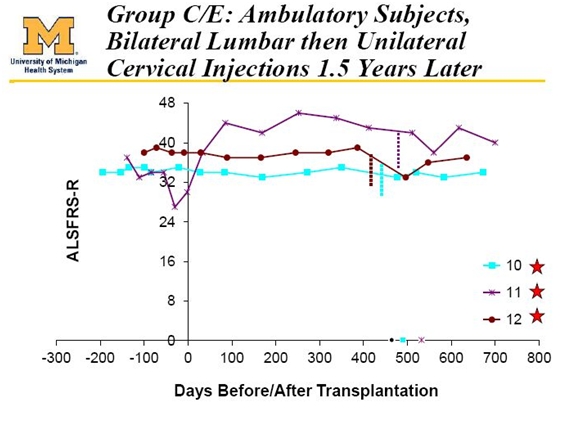
The Most Interesting Cohort is Cohort C/E
The next three patients, patients 10, 11 and 12 were all ambulatory and initially received ten bilateral lumbar injections. Remember that patients 7, 8 and 9 received five unilateral injections in the lumbar region so that 10, 11 and 12 received twice as many cells. These three patients then received a second transplantation one and one half years later of five unilateral injections in the cervical region.
These three patients are the most meaningful of the total of 15 patients treated. They received the highest number of injections and had the best results; the results are quite impressive.
Ted Harrada is patient 11 and his ALSFRS-r score improved dramatically from 31 when he entered the study to a high of about 46 (remember 48 is normal) and he is now at 40 after two years. Under the rule of thumb that ALSFRS-r score declines 0.9 points per month, we might have expected his score to be 13 and for him to be near death. Dr. Feldman and other KOLs have never seen an improvement like this before. ALS patients just don’t spontaneously improve; their decline is inexorable.
While all of the attention has been focused on Ted Harrada, patients 10 and 12 have been stable for two years and this is almost as impressive.
The data above shows patients 10, 11 and 12 data going out about 700 days. I would note that the slides were two months old and that at this stage the patients only come back for a checkup every six months or 180 days. Hence, the actual time from transplantation could be anywhere from 60 to 240 days longer than shown in the slide. As far as I know, these patients continue to do well.
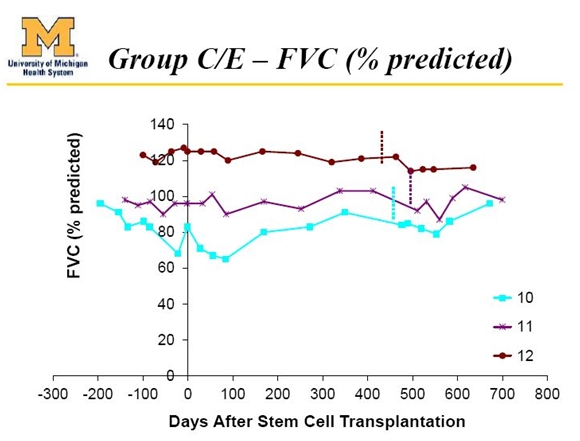
Most patients with ALS die from respiratory complications resulting from a loss of function in nerves in the cervical region of the spine that control muscles that control respiratory function. If these stem cells are going to prolong life, it will be because they slow the progression of this muscle weakness. Dr. Feldman presented a slide on FVC that was quite encouraging.
The FVC or forced vital capacity test requires the patient to take the deepest breath they can and then exhale into a sensor as hard as possible, for as long as possible, preferably at least 6 seconds. This is a standard test for measuring respiratory function. The results are shown below:
These results are probably more meaningful than ALSFRS-r because maintenance of respiratory function is essential to prolonging life. You can see from the above slide that Ted Harrada’ s (patient 11) breathing function has been maintained and patients 10 and 12 are also stable.
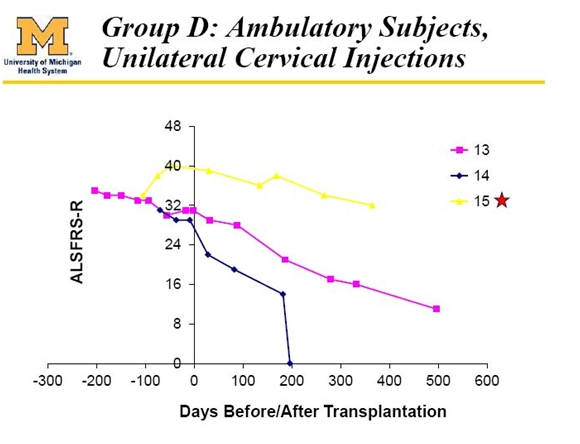
Cohort D
Three patients in group D differed from all others in the trial as they presented with bulbar symptoms which are due to deterioration in nerves in the cervical regions of the spine. These nerves control functions such as breathing, eating and speech. ALS symptoms usually start with deterioration of nerves in the lumbar region that then progress over time to the cervical region. Patients with bulbar symptoms have a different course of disease progression that is more rapid for reasons that are not well understood.
It can be seen that patients 13 and 14 had a rapid deterioration in their condition while patient 15 appeared to do somewhat better. Dr. Feldman is pessimistic that the treatment will be effective in ALS patients that present first with bulbar symptoms. They will be excluded from the phase 2 trial.
Conclusions and Investment Significance
I think that the transplantation of cells in patients 1, 2, 3, 4, 5 and 6 in cohort A provided no benefit. I don’t think that any benefit should have been expected as these were very advanced patients who received only five injections.
The poor results of patients 13 and 14 in Cohort D indicate that the cells will probably not produce any benefit in patients who present with bulbar symptoms. Such patients will be excluded from phase 2.
Most investor attention has been focused on the results of Ted Harrada and the other two patients in Cohort C/E. They received the most injections (15) and their results were the best. Neuralstem feels that even so, patients 10, 11 and 12 were given sub-optimal numbers of cells.
I think that the most surprising finding for me in this presentation is how well patients 7 and 8 fared in Cohort B. These patients received only five injections in the lumbar region as opposed to Ted Harrada and patients 10 and 12 in Cohort C/E who received 10 bilateral injections in the lumbar region and later five injections in the cervical region. Patients 7 and 8 received only one-third the numbers of cells and while they did not fare as well, the rate of decline in their disease seems to have been slowed.
In interpreting these results, I think that we can conclude that the lack of efficacy in patients 1,2,3,4, 5 and 6 was because their disease was very advanced and they received only a small number of cells. I think that poor results in patients 13 and 14 may show that the transplantation is ineffective in bulbar disease although I note that they received only five injections.
In searching for signals of efficacy, clearly the results in patients 11 (Ted Harrada) 10 and 12 are remarkable. Ordinarily, I would ignore results based on this small a number of patients, but this is ALS. In this condition, we see all patients experience an inexorable decline in neurological function and ultimately death. These three patients have shown stability or just a gradual decline in their disease over a period of 700 or more days and this is extraordinary for the general ALS population.
While the results in patients 7 and 8 were less dramatic, they are still encouraging signals of efficacy. I have previously explained why I think that patients 10, 11 and 12 in Cohort C/E and potentially patients 7 and 8 from Cohort B would be most likely to benefit. It appears that all of these patients or five out of five who would be expected to benefit did show signals of efficacy. I find this to be extremely encouraging and is the basis of my recommendation. The patient numbers are uncomfortably small but the results just can’t be dismissed as occurring by chance.
Phase 2 Trial is Underway
Dr. Feldman did not have much to say about the phase 2 trial in terms of efficacy. She said that 11 patients have been entered in the trial. She presented no sides on efficacy but noted that as in phase 1 there were no safety concerns and all patients are doing well. The general design of the trial is shown below:
The important thing to note in this phase 2 study is that the first 11 patients have received only cervical injections. Clearly, the study is trying to improve the functioning of muscles that control respiratory function that are controlled by nerves originating in the cervical region.
It is also important to note the much greater number of cells being injected. Ted Harrada, patient 11 and patient 12 received the most number of cells in the phase 1 study with 1.5 million cells. Phase 2 started with 2 million cells and the patients 10 and 11 in the study have received 8 million cells. The final cohort will receive 16 million cells. Based on animal studies, Neuralstem believes that the optimal number of cells could be more than 16 million.
A final group of three will be comprised of three patients who have previously received cervical injections and will be brought back for lumbar injections. This will complete enrollment. There is a six month follow-up for each patient so that the topline results for the entire trial are probable for very late 4Q, 2014 or early 2015. However, because this is an open label trial we could see results for individual patients earlier than this. Assuming success, the phase 3 trial could start in 1H, 2015.
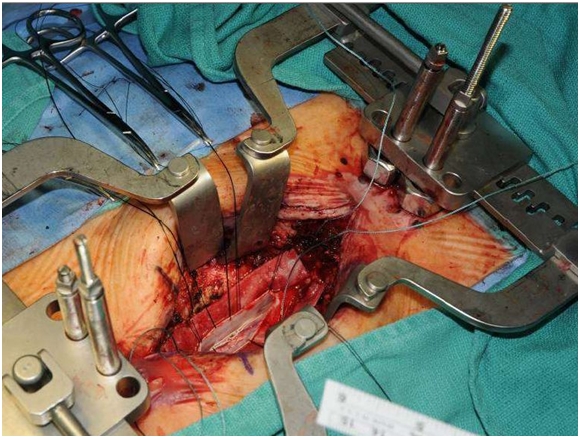
The Surgery
The surgery is not a simple procedure. These cells are injected directly into the spinal column and the surgery exposes the spinal column as shown below:
The spinal column is moving during the procedure as the patient breathes, which presents an issue. If the injection is made incorrectly because of this movement, it could cause damage to the spine. Neuralstem has rights to a surgical device that allows the injection to be synchronous with the movement of the spine. This is an extremely important invention that is shown below:
Refresher on ALSFRS-r Scale
ALSFRS-r is a scale that measures the decline in bodily function during the course of ALS. This is done by a physician assessing 12 different functions and assigning a value of 4 (normal) to 0 (function is absent). A normal person would score 48. The components of the scale are shown below:
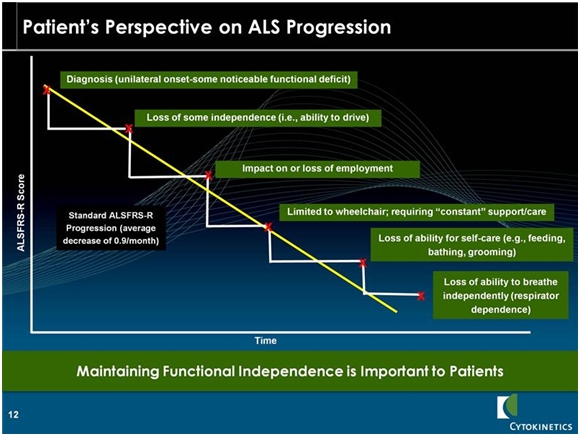
At the time of diagnosis most patients have a score of around 36. Historical data shows an inexorable decline in the ALSFRS-r scale of 0.9 to 1.0 points per month. Patients usually die when the ALSFRS-r score declines to the high single digits. The key opinion leaders I have listened to say that occasionally patients may stabilize for a month or two, but the decline in ALSFRS-r is inexorable. What this does to patients is graphically illustrated in the following chart.
Tagged as ALS, amyotrophic lateral sclerosis, CUR, Inc., Lou Gehrig's disease, Neuralstem, Inc. + Categorized as Company Reports
5 Comments
Trackbacks & Pingbacks
-
Neuralstem: Investigators are Very Positive on NSI-566 Results in ALS (CUR, Buy, $3.27) | Expert Financial Analysis and Reporting | Smith on Stocks
[…] Patients usually die about three to five years after diagnosis when their ALSFRS-r score is 12 to 18. Dr. Glass is talking about these three patients being stable at three years when the expectation would be that they would be nearing death at that point. For a detailed description of the patients in the phase 1 trial please follow this link. […]
-
Neuralstem: A Closer Look at Encouraging Phase 2 Results for NSI-566 Neural Stem Cells in Treating ALS (CUR, Buy, $2.36) | Expert Financial Analysis and Reporting | Smith on Stocks
[…] in the lower spine. A detailed discussion of the design and results of this trial can be seen in my report of April 11, 2014. As explained in that report, three of the first six patients treated were advanced patients who […]
Comment
You must be logged in, or you must subscribe to post a comment.








Larry:
I don’t know how you handle these comments or if cannot/prefer not to respond or there is a way that you do respond.
I left a comments a couple weeks ago regarding the data presented that inquired about: 1) the second cohort who had higher initial ALSFS scores but were to ambulatory, what seemed a contradiction, and 2) the last cohort who are identified to all be bulbar, which I wasn’t sure was the case.
I have a couple further comments: 1) there are a couple new Phase II patients with blogs but they are family/friend blogs and so I don’t feel comfortable broadcasting their links. I assume you have already heard of them. Nevertheless, one seems to show no progression, but no improvement, six months subsequent to surgery while the other doesn’t discuss symptoms but does discuss the invasiveness of the surgery. I guess this first patient is one more indication of a patient with stability post surgery.
Last the experience with CYTK goes to show how initial superficial data can be misleading. I’m not sure if we have the same circumstance here with CUR as the preponderance of our small data set seems to show something is going on, but it is certainly a treacherous investment world out there.
Thanks for the comment.I encourage people to be active on the comments section.
In group D or patients 13, 14 and 15, I am certain that patients 13 and 14 presented with bulbar symptoms and I think that patient 15 also had bulbar symptoms. This was the purpose of Cohort D to look at patients presenting with bulbar symptoms. However, patient 15 shows a different response without the steep decline in ALSFRS-r of the other two. I will check with the Company on this.
Stability for six months for the person in phase 2 is considered remarkable. Even patients 10, 11 and 12 who have shown the most improvement are pretty much stable but for a longer time frame.
Biotechnology can be a wicked siren as our ability to project results in larger phase 2b or phase 3 trials from smaller phase 2a and phase 1 trials is limited. I was swayed by the scatter plots of patients in the phase 2a trials and BENEFIT patients enrolled prior to November 2013. I was not alone. The lead investigator for the trials who is one of the key opinion leaders in ALS was also optimistic that we were seeing a biological effect. Perhaps with the detailed analysis that will be done on BENEFIT we will learn a lot more. ALS is a treacherous disease in which no drugs have been successfully developed for 20 years despite numerous efforts.
There were only five patients in the CUR phase 1 trial who could have expected a benefit and all five have shown stability over a remarkable period of time and we seem to see this with the two patients you referred to from phase 2. The results are extremely encouraging, but they are only seven patients.
Mr Smith: After re-reading this report I have a question about the Phase II trial. Given that the patients are relatively early phase, non-bulbar, and ambulatory coupled with groups A-D receiving only cervical injections could the impact of NSI-566 on the ALSFRS-r scale be muted at the time of the six month reviews?
My non-medical understanding gained from your writing is that non-bulbar ALS starts in the extremities and then progresses to the core in later stages. While I also understand that the benefit of NSI-566 may extend beyond the area directly treated, I am concerned that the study design might require a long period of tracking to show benefit. That is, could we expect that with the lack of lumbar injections that these patients’ ability to walk will continue to decline while their breathing capabilities would not yet get to the point where they would begin to experience the decline expected over the “normal” course of the disease?
If this is true, the six month followups may not show much benefit, even if NSI-566 is working exactly as expected? If that is the case, time would reveal that to be so, but couldn’t this prove to be a vulnerability for investors? Now, it would seem that CUR is not following the NWBO formula of providing ongoing updates (it would seem likely that at least a few of the 11 treated patients as of April have had their six month followup appointments) so the company could just sit tight throughout this period. Still, you have stated that you are expecting top-line results by early 2015 as presumably are other professional investors.
My concern would be completely unfounded if each of the 12 components for ALSFRS-r decline at a relatively uniform rate. Thank you.